AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
K and ca element12/17/2023  What is the electronic configuration of Calcium 20? What is the boiling Point of Calcium in Kelvin?īoiling Point of Calcium in Kelvin is 1757 K. Melting Point of Calcium in Kelvin is 1115 K. What is the melting Point of Calcium in Kelvin? What is the boiling Point of Calcium?īoiling Point of Calcium is 1757 K. Calcium has 20 electrons out of which 2 valence electrons are present in the 4s2 outer orbitals of atom. How many valence electrons does a Calcium atom have?Ĭalcium has 2 valence electrons. What is the color of Calcium?Ĭalcium is of Silver color. It is located in group 2 and period 4 in the modern periodic table. Calcium is the 20 element on the periodic table. What is the position of Calcium in the Periodic Table?Ĭalcium is a chemical element with the symbol Ca and atomic number 20. Calcium is a chemical element with symbol Ca and atomic number 20. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Calcium is 4s2. What is the abbreviated electronic configuration of Calcium? 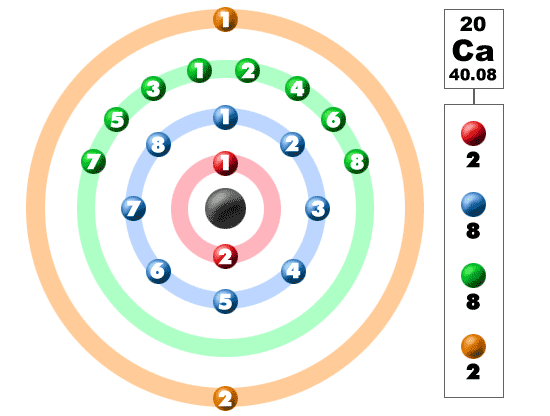
The electronic configuration of Calcium is 1s2 2s2 2p6 3s2 3p6 4s2. What is the electronic configuration of Calcium? Optical Properties of Calcium Refractive IndexĪcoustic Properties of Calcium Speed of SoundĬalcium Thermal Properties - Enthalpies and thermodynamics Refer to table below for the Electrical properties ofCalcium Electrical ConductivityĬalcium Heat and Conduction Properties Thermal ConductivityĬalcium Magnetic Properties Magnetic Type Hardness of Calcium - Tests to Measure of Hardness of Element Mohs HardnessĬalcium is Conductor of electricity. At 3.5 oxygen is a distant second, while chlorine and nitrogen are tied for third place at 3.0.Refer to below table for Calcium Physical Properties Densityġ.55 g/cm3(when liquid at m.p density is $1.378 g/cm3) Fluorine, with an electronegativity of 4, is by far the most electronegative element. Another feature worth noting is the very large differences in electronegativities in the top right-hand comer of the table. Between these two extremes we notice that most of the remaining metals (largely transition metals) have electronegativities between 1.4 and 1.9 (light gray), while most of the remaining nonmetals have electronegativities between 2.0 and 2.4 (light red). They are often referred to as the most electropositive elements, and they are the metals which invariably form binary ionic compounds. These elements have been coded in dark gray. By contrast, elements with negativities of 1.3 or less are all metals on the lower left of the table. Periodic Table → Atomic radius decreases → Ionization energy increases → Electronegativity increases →Īs can be seen from this table, elements with electronegativities of 2.5 or more are all nonmetals in the top right-hand comer of the periodic table. In this scale a value of 4.0 is arbitrarily given to the most electronegative element, fluorine, and the other electronegativities are scaled relative to this value. 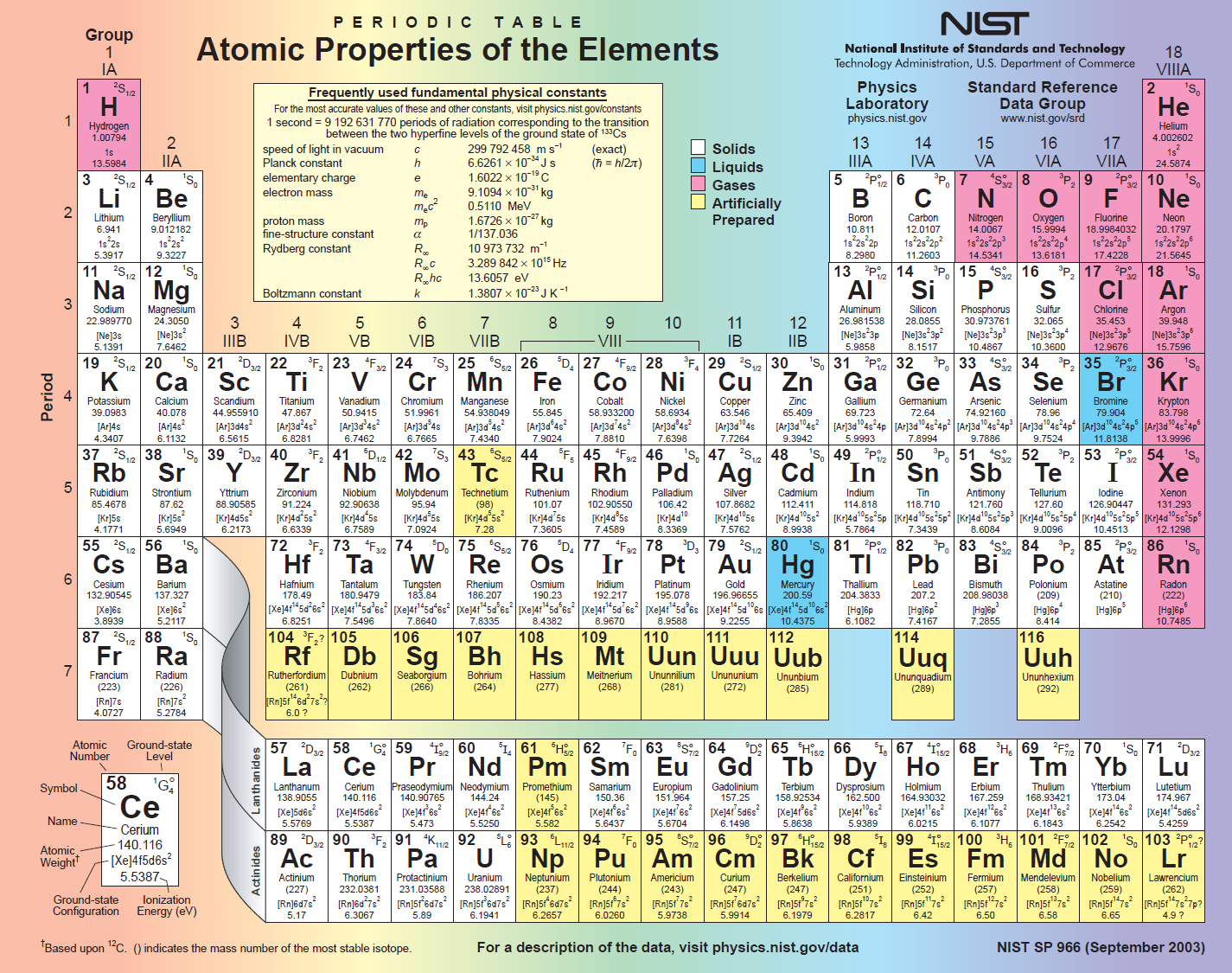
The best-known of these scales was devised by the Nobel prize-winning California chemist Linus Pauling (1901 to 1994) and is shown in the periodic table found below. Nevertheless most of these attempts agree in large measure in telling us which elements are more electronegative than others. Various attempts have been made over the years to derive a scale of electronegativities for the elements, none of which is entirely satisfactory. Therefore the dipole moment cannot tell us quantitatively the difference between the electronegativities of two bonded atoms. 
Also, the polarity of a bond depends on whether the bond is a single, double, or triple bond and on what the other atoms and electron pairs in a molecule are. Dipole-moment measurements tell us about the electrical behavior of all electron pairs in the molecule, not just the bonding pair in which we are interested. Unfortunately there is no direct way of measuring electronegativity. Furthermore the more polar a bond, the larger the difference in electronegativity of the two atoms forming it. The negative side of a polar covalent bond corresponds to the more electronegative element. The ability of an atom in a molecule to attract a shared electron pair to itself, forming a polar covalent bond, is called its electronegativity. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
